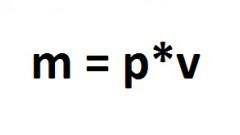You will need
- A pen and a piece of paper
Instruction
1
Find reference information about the measured product and write down the value of its density.
2
3
The mass of a substance is split into a reference density value. The result of the action will correspond to the volume of product in cubic meters.
4
Translate cubic meters to milliliters. To do this, the result of previous actions multiply by 1000000. The result is our desired value is the volume in milliliters for this product. You can then measure in a measuring Cup specified quantity.
Note
The density of the product should be given in the reference in the SI metric system, i.e. it is specified in units of kg/m3.
Useful advice
Sometimes in a table reference point is not the exact number, but the range of density values, such as buckwheat 1180-1280 kg/m3. In this case, you should take the average value of these values, as it is closest to the density given in less precise tables.