Experiment
To determine the mass of 1 l of distilled water you can use a fairly simple experiment. For it will need:
- distilled water;
- measuring utensils;
- glass or plastic Bank;
- electronic scales.
Distilled water you can buy in the pharmacy or in a shop for motorists. Measuring utensils are quite often sold in hardware stores, and electronic scales. Weigh the jar, write down its mass. Pour into a measuring bowl with exactly 1 liter of water. Pour water into the jar, weigh. Subtract the mass of the banks. You will get the result close to 1 kg. the Most accurate weighing is obtained at a temperature of 4°C and a pressure of 760 mm Hg. Under these conditions, 1 l of distilled water has a mass of 998,5 G.
Since aggressive substances, you will not run, chemical ware in this case is not mandatory.
The water is a different
Spend the same experiment with ordinary tap water. Similarly, weigh the jar, fill it with water, pre-measure volume with volumetric glassware. The result will differ, although not by much. In tap water may contain impurities heavier substances, so 1 liter of water may weigh a little more than the same amount of distillate. Heavy water you are unlikely to get it, besides experimenting with it is definitely not worth it. The mass of 1 liter of such water will be much greater than 1 kg.
No experiments
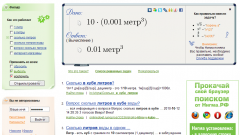
Calculate the mass of 1 liter of water and no experiments. Remember, what is equal to 1 liter Is 1 cubic. DM, that is 1000 cubic cm the Density of distilled water is 1 g/CC Mass m equal to the density ρ multiplied by the volume V. That is to calculate it by the formula m=V*ρ=1000 CC*1G/CC=1000 g
Other volumes
In life sometimes you need to determine the mass of another volume of water. For example, bucket or Cup. It is important to know how much liquid this holds capacity. The standard glass usually includes 250 ml of water, i.e. ¼ L. in Order to calculate the mass of the water, just 1 kilograms divided by 4. You will receive approximately 250 g. If you weigh more accurately, the mass of water will amount to $ 249.6 m in Order to determine how much weight a bucket of water, you also need to know how much liquid is included in this bucket. For example, the number of ordinary tap water which is placed in a 10 litre bucket weighs about 10 kg, 12-liter – 12 kg etc.
Other liquids
With the help of scales you can learn, for example, what is the weight of a liter of milk. Since the average density of milk the density of water, l will weigh a little more than a kilogram.
In the case of milk, you can only talk about the average density, since milk is a mixture of various substances.As for other liquids, it is best to calculate their weight according to the formula as it is not always possible to weigh the container. In addition, fluid can be aggressive to work with in the home in the absence of special chemical glassware is unsafe, the density of acid and alkali can be found in the table. You will only have to determine the volume of the vessel and to substitute the numbers into the formula.