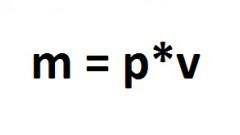You will need
- calculator;
- - handle;
- table physico-chemical properties of petroleum products;
- paper.
Instruction
1
In order to transfer a literand of gasoline , in kilograms, calculate the weight of fuel, that is, its density. This value has special significance for the storage of fuel in the tanks. The density of gasoline can be found by dividing its mass by the volume. However, the density of all petroleum products is relative, since so much depends on changes in air temperature.
2
Dealing with a dimensionless quantity, divide the volume of gasoline at a temperature, which was recorded at the time of determining the mass of water at a temperature of + 4oC, taken in exactly the same volume, because at this temperature it is known that the mass of water is one kilogram. The resulting value is the relative density.
3
With increasing temperature the volume of gasoline increases. To determine the density, subtract from the initial high temperature and resulting value multiply by the coefficient of volume expansion of gasoline. The value of the coefficient in the table find the physico-chemical properties of petroleum products. To the desired product of the add unit. The resulting amount and multiply by the original amount of gasoline. Thus, you will learn how to change the volume of gasoline with increasing temperature.
4
To put a litre of petrol in pounds, divide its volume by the density. Average values of density, independent of temperature, regulated by the state and approved by the Ministry of industry and energy.
Note
In order to avoid errors when writing to registers, keep records of gasoline in two natural units of measurement – liters and kilograms.
Useful advice
In order to avoid becoming a victim of fraud vendors often overstate the actual density, and hence the price, it is advisable to keep records of gasoline in liters.