Instruction
1
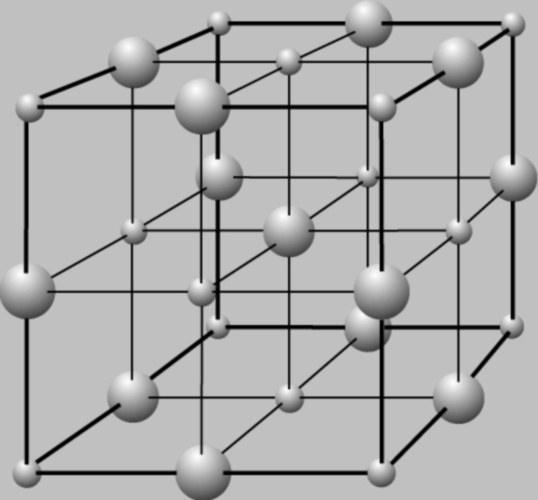
As you can easily guess from the name, the metal lattice type found in metals. These substances are characterized by generally high melting point, metallic luster, hardness, are good conductors of electric current. Remember that the nodes in lattices of this type are either neutral atoms or positive ions. In between nodes the electron migration which provides a high conductivity similar substances.
2
Ionic crystal lattice type. Keep in mind that it is inherent in the oxides and salts. Typical example – all known crystals of table salt, sodium chloride. The nodes of such gratings alternately alternating positively and negatively charged ions. Such substances are generally refractory, with low volatility. As you can guess, they have an ionic type of chemical bonding.
3
The atomic type of the crystal lattice inherent in simple substances, non – metals, which under normal conditions represent a rigid body. For example, sulphur, phosphorus, carbon. The nodes of these lattices are neutral atoms, connected to each other via a covalent chemical bond. Such substances characteristic of refractoriness, insolubility in water. Some (for example, carbon in form of diamond) – extremely high hardness.
4
Finally, the last type of lattice - molecular. It is found in substances under normal conditions are in liquid or gaseous form. As, again, you can easily understand from the name, the nodes of these lattices, molecules. They can be as non-polar species (from simple gases such as Cl2, O2) and polar species (the most famous example – water H2O). Substances with this type of lattice does not conduct electricity, volatile, have low melting temperatures.
5
Thus, to confidently determine what type of crystal lattice has a particular substance, you ought to find out to what class of substances it is and what physico-chemical properties of has.