Instruction
1
In redox reactions, some elements act as donors of electrons, i.e., oxidize; others as acceptors, i.e., restored.
2
In cases of interaction of typical oxidizing and reducing agents you can immediately identify what we are talking about redox reactions. For example, the interaction of alkali metals with acids or Halogens, combustion in oxygen.
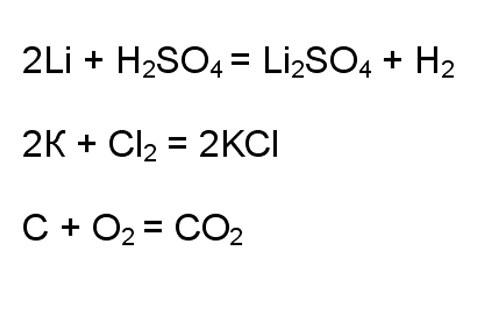
3
Let's consider a more complicated case, for example, reaction of potassium permanganate with potassium sulfite in the presence of a large number of alkali CON. To make sure the reaction is redox, identify the oxidation numbers of the elements in the right and left side. Atoms of one element always take or give the same number of electrons. In this reaction are oxygen, hydrogen, potassium. The other oxidation state can be different for manganese and sulfur.
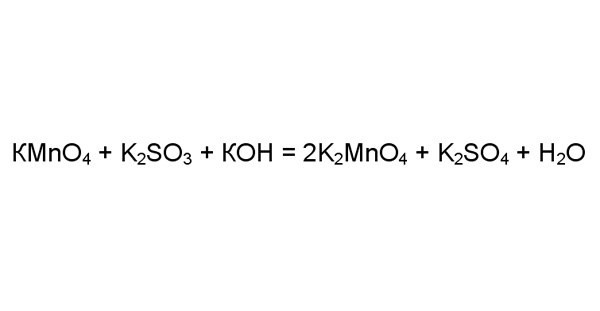
4
Determine the oxidation state of manganese and sulfur on the left side of the equation. Take the potassium permanganate oxygen is always the acceptor of electrons the oxidation number (-2). Four attached oxygen atom 8 electrons. Potassium is an electron donor, and its oxidation state (+1). One potassium atom gives up one electron. Then manganese must give: 8-1=7 electrons.
5
Similarly, determine that the oxidation number of sulphur in sulphide of potassium (+4). Three atoms of oxygen take 6 electrons, two atoms of potassium to give two electrons.
6
Now, find the oxidation States for these elements in the right part. In manganate K2MnO4 potassium four oxygen atoms attached to eight electrons, two atoms of potassium give two. Hence, the reduced manganese oxidation state (+7) (+6), i.e. recovered.
7
The sulfur in potassium sulfate, on the contrary, oxidized (+4) (+6). In the K2SO4 molecule of four atoms of oxygen take eight electrons, two atoms of potassium give two. Therefore, six electrons are taken from the sulfur atom.
8
Oxidation of manganese and sulphur has changed. And you can conclude that this reaction is redox.
